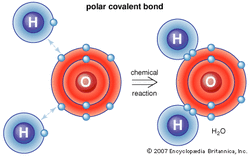
The chemical symbol for water is H2o, or 2 hydrogen atoms and 1 Oxygen atom. Atoms are the smallest unit of matter that retains its properties, and it consists of: negatively (-) charged electrons orbiting a central nucleus, made of protons (+) and Neutrons (neutrally charged particle).
One property of water is it's strong Polar/ Covalent bonds. Polarity refers to the unequal sharing of electrons. These bonds are caused by the pull between the hydrogen and oxygen atoms. This pull is caused by the fact that the Oxygen atom is able to attract much more electrons than the Hydrogen atom is, due to size and mass. (See Left)
One property of water is it's strong Polar/ Covalent bonds. Polarity refers to the unequal sharing of electrons. These bonds are caused by the pull between the hydrogen and oxygen atoms. This pull is caused by the fact that the Oxygen atom is able to attract much more electrons than the Hydrogen atom is, due to size and mass. (See Left)
- Another property of water is its ability to be a Universal Solvent. This means that in a lot of reactions, water can dissolve many substances.
- Cohesion is a property of water that has to do with its tendency to stick to itself. (see top-left picture). This also has a lot to do with Hydrogen Bonds, but we'll talk about that later.
- Adhesion is water's tendency to stick to other materials and objects, such as leaves (see bottom-left)
- Surface Tension is when the weak, magnetic-like hydrogen bonds in the water cause a thin, film-like layer at the surface of any amount of still water.

- Surface Tension is when the weak, magnetic-like hydrogen bonds in the water cause a thin, film-like layer at the surface of any amount of still water, allowing very lightweight objects (like this bug) to "stride" the surface of the water without falling in
- Another property of water is its Capillary Action. This property allows water to use both cohesion and adhesion to move upwards, against the force of gravity (see picture of Blue Daisy)
- Another property of water is its High Heat of Vaporization. This means that water can absorb a high amount of heat before evaporating. This is how Sweating cools you off

- Another property of water is its High Heat of Vaporization. This means that water can absorb a high amount of heat before evaporating. This is how Sweating cools you off
- High Latent Heat of Fusion: This is a property of water that means that a lot of heat is released when water freezes and a lot of heat is absorbed when water evaporates.
- Last, but not least, water has a very high specific heat. This means that water can absorb a large amount of heat (see button)
