Marine Science: Intro To Density Lab All About Layers!

First, we added blue food coloring and yellow food coloring to 2 different beakers filled with 250 ml. of water, with the yellow beaker containing warm, fresh water and the blue one containing cold, blue water with 20g of salt added and dissolved in the water.
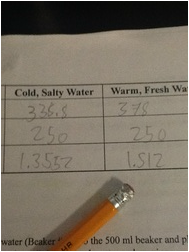
Then, we calculated the density of the water in the beakers, discovering that the warm, fresh water was less dense, at about 1.35 grams per cubic milliliter, than the cold, salty water, at 1.512 grams per cubic milliliter.

Lastly, we emptied both beakers into a larger, 500 ml. beaker. Eventually the water mixed to become green, but for a short time, the yellow water stayed at the top and the blue water at the bottom. This demonstrates the principle of stratification, or how the ocean is divided into regions of greater and lesser density (due to the salinity of the water), separated by a thermocline. It often mixes because of wind, welling and storms. This causes nutrients to be spread throughout the ocean, so that life can exist on all levels of the sea.
